









| Technical Specification | |
|---|---|
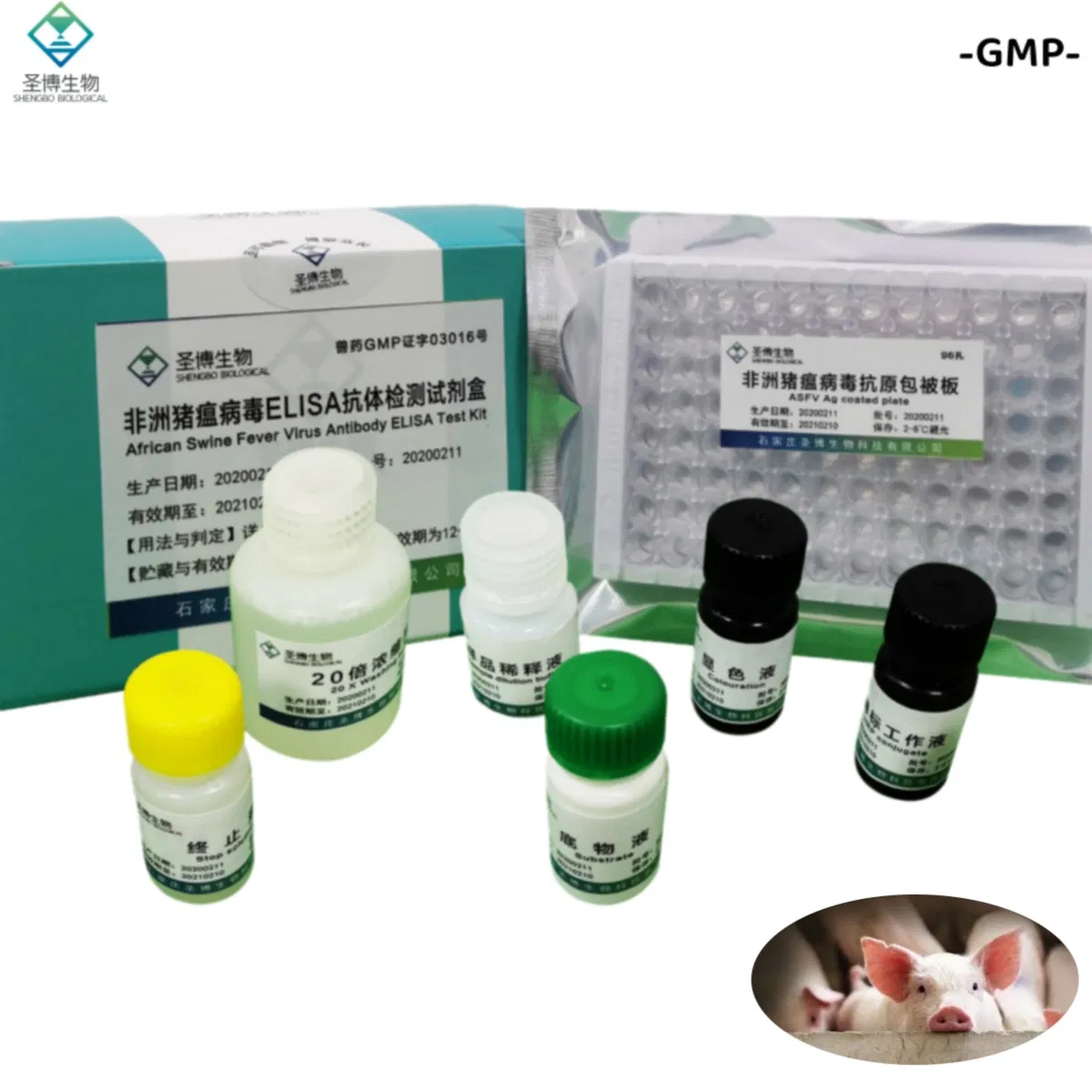
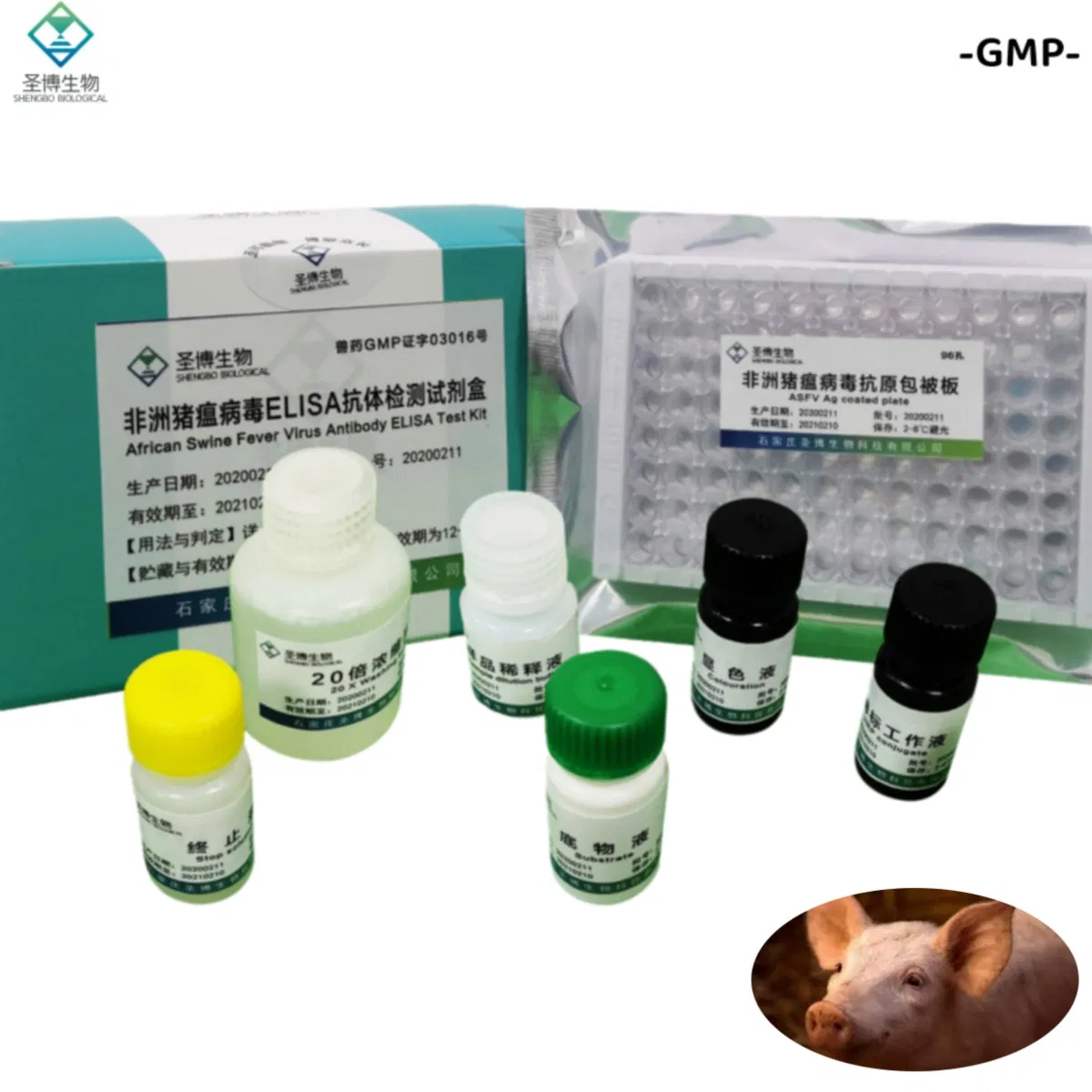
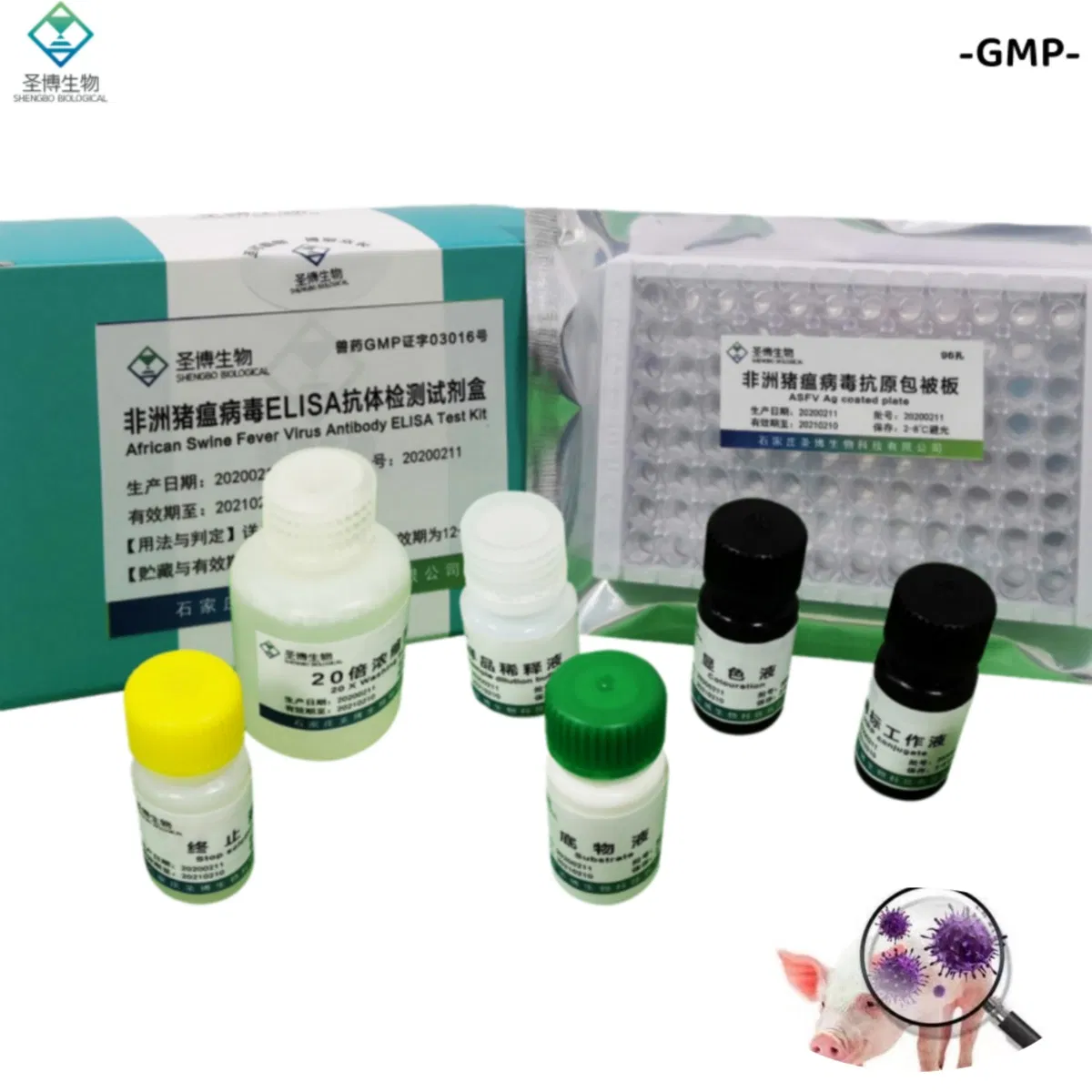
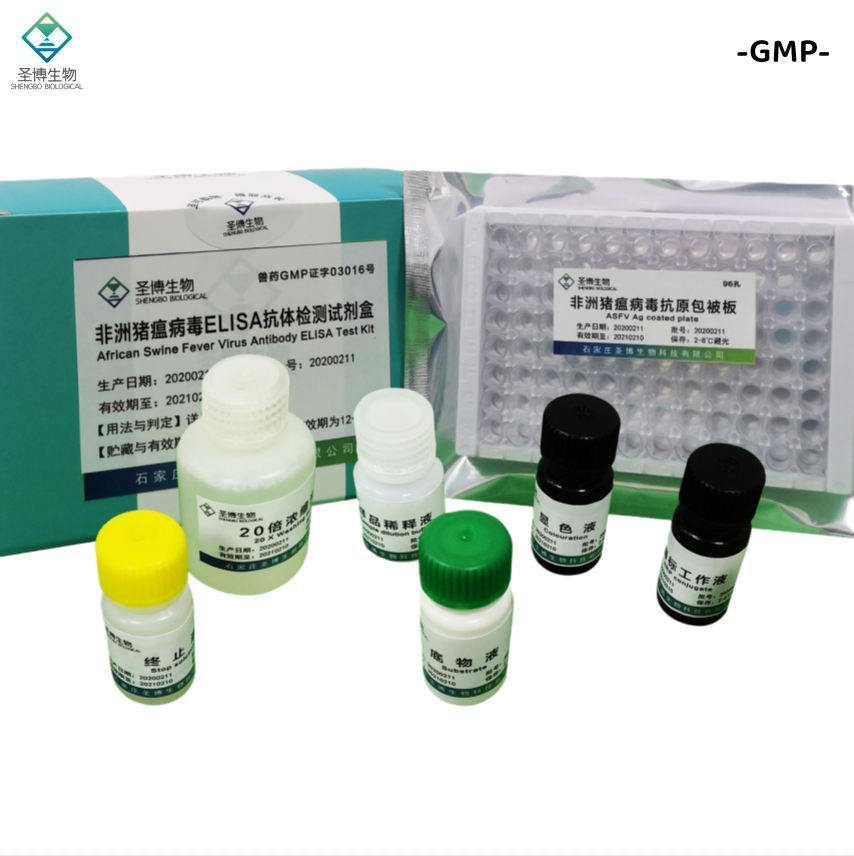
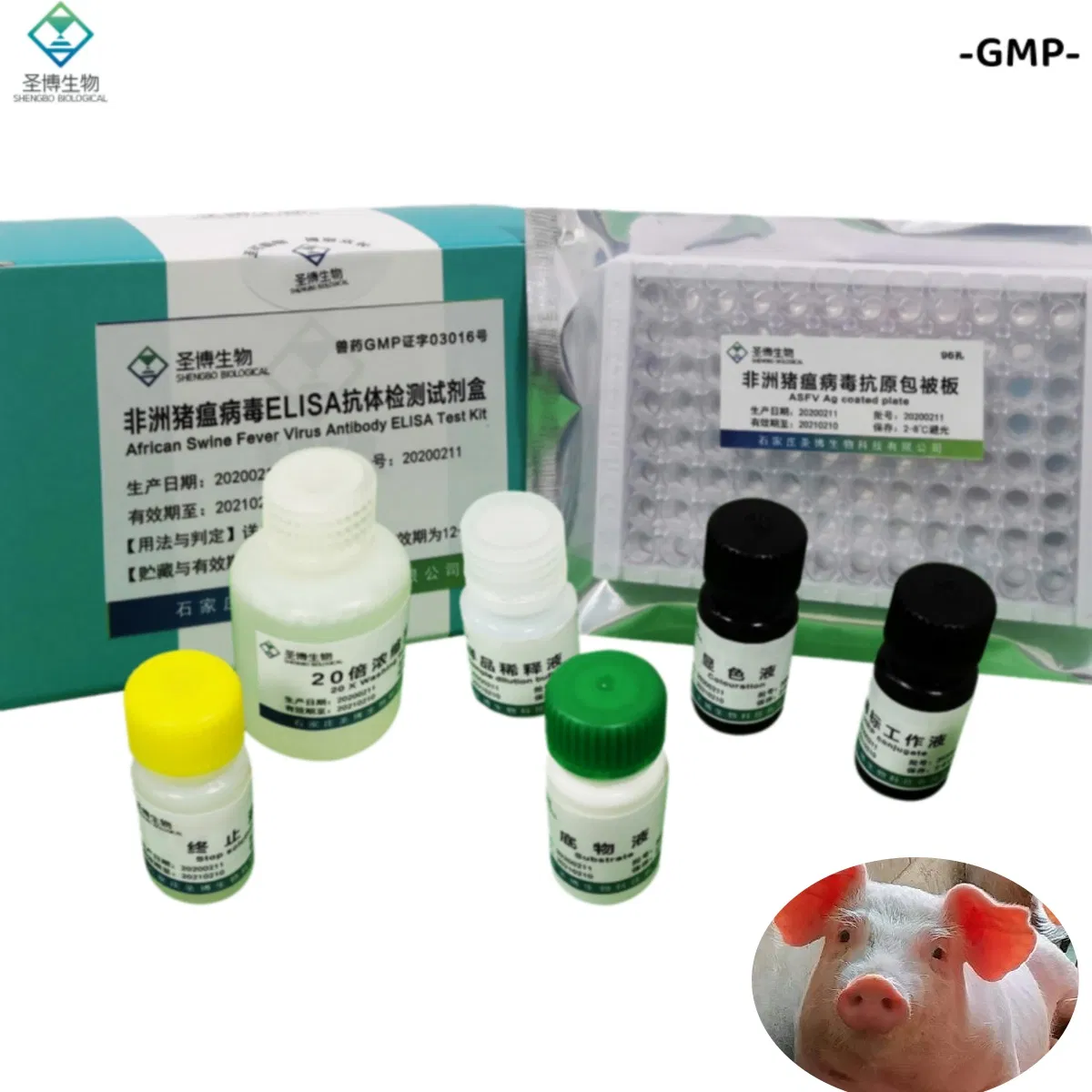
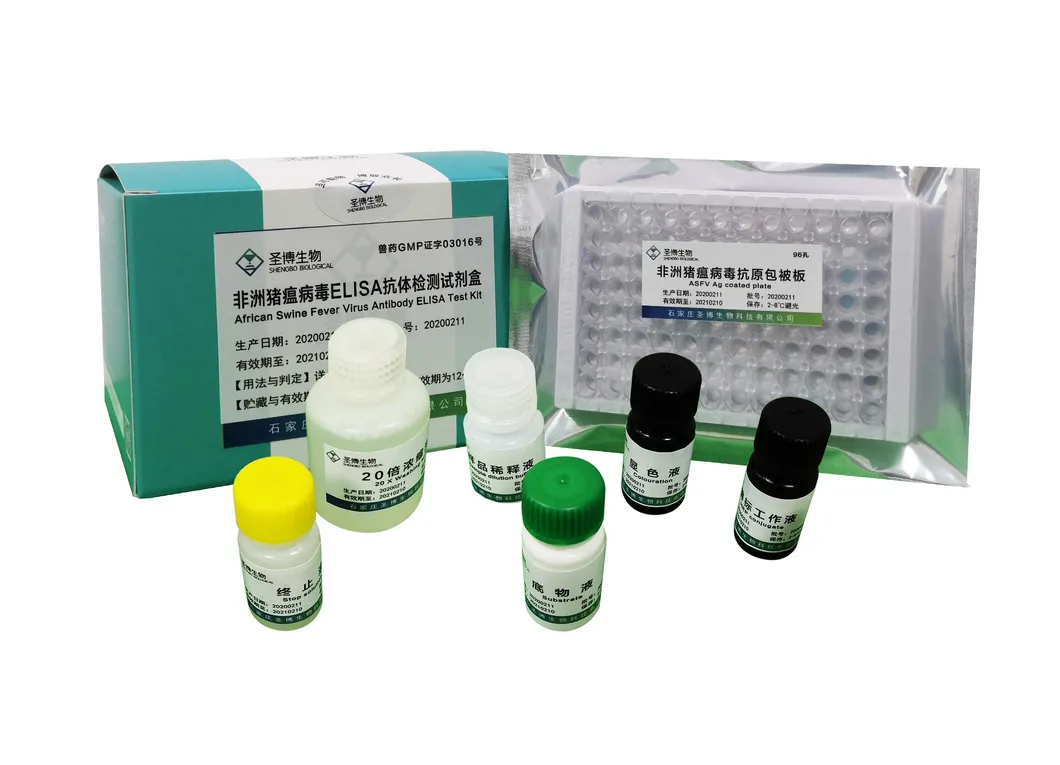
| Name | African Swine Fever Virus Antibody ELISA Test Kit |
| Package | 96 T/Box |
| Storage Condition | 2~8ºC |
The product consists of a microplate coated with genetically engineered ASFV recombinant antigen, HRP-labeled antibody, and other essential reagents. It utilizes the principle of indirect enzyme-linked immunosorbent assay (ELISA) to detect ASFV antibodies in pig serum or plasma. It is primarily used for the auxiliary diagnosis of African swine fever.
| No. | Component Name | Quantity (96T×1) | Quantity (96T×2) |
|---|---|---|---|
| 1 | Enzyme working reference solution | 11mL | 22mL |
| 2 | Sample diluted solution | 11mL | 22mL |
| 3 | Positive reference serum | 1mL | 2mL |
| 4 | Negative reference serum | 1mL | 2mL |
| 5 | Substrate solution | 6mL | 12mL |
| 6 | Color solution | 6mL | 12mL |
| 7 | Stop solution | 6mL | 12mL |
| 8 | 20× concentrated lotion | 50mL | 100mL |
| 9 | Instruction Manual | 1 piece | 1 piece |
| 10 | Sealing film | 3 pieces | 6 pieces |


The facility covers over 26,000 square meters with a construction area of 11,000 square meters. Featuring 11 workshops and 14 production lines that have received formal GMP certification. As a leading manufacturer of prepared drugs, the products are widely recognized in domestic and international markets across America, Africa, Europe, and Asia.









Participated in VIV Asia, AgriTek Astana, and EuroTier 2022

