Basic Information
🏷️
Logo Printing
OEM Brand
✔️
Advantage
on-Site Drug Testing
⚙️
Feature
One-Step Operation
📦
Transport Package
Carton or Box
📊
Production Capacity
50000/Day

HIV-1/2 Antibody Urine Assay Kit (Colloidal Gold)

📍 Intended Use
This product is used for the qualitative determination of human immunodeficiency virus type I (HIV-1) antibodies and type II (HIV-2) antibodies in human urine samples. It is suitable for the auxiliary diagnosis of HIV infection. The test results are only for clinical reference and cannot be used alone as the basis for confirming or excluding cases. This product can be used for consumer self-test.
Product Specification
| Parameter |
Details |
| Product Name |
HIV Antibody Urine Test kit (Colloidal Gold Method) |
| Method of Detection |
Colloidal Gold Method |
| Qualitative/Quantitative |
Qualitative |
| Specimen |
Urine |
| Required Volume |
3 drops (approximately 100ul) |
| Packaging |
Cassette: 1 test/box, 10 tests/box, 20 tests/box, 50 tests/box. |
Storage & Validity
1. Store at 4~30ºC, avoid light and dry, valid for 24 months.
2. See the outer packaging for the production date and expiration date. Do not freeze.
3. After opening the sealed pouch, use the test as soon as possible within 1 hour.
Testing Procedure
Allow the test box to equilibrate to room temperature prior to testing.
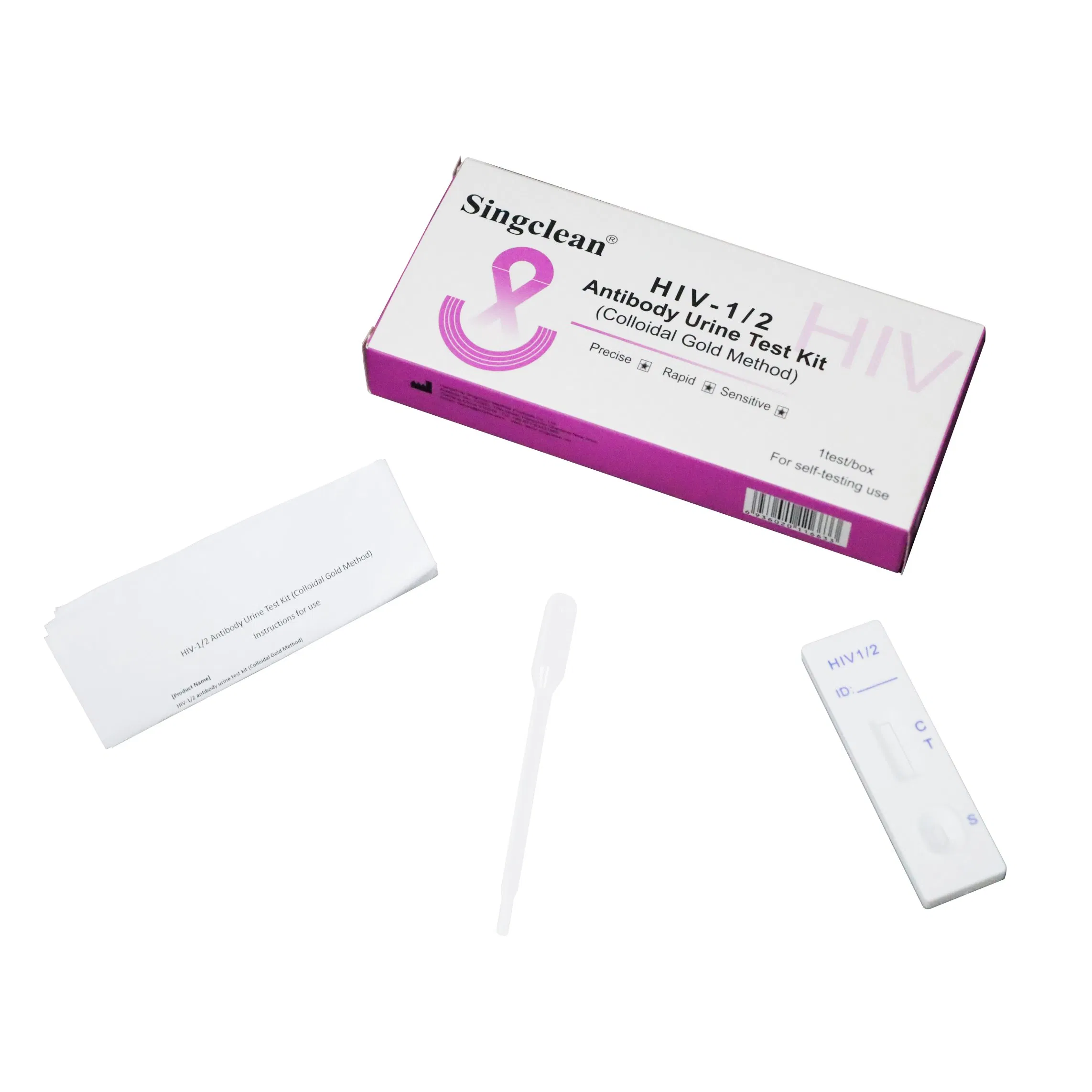
Step 1: Remove the test card from the sealed foil bag and use it within 1 hour.
Step 2: Place the test apparatus on a clean, level surface.
Step 3: Aspirate the urine sample with the provided dropper, add 3 drops (100ul) to the sample well.
Step 4: Read results after 15 minutes. Results after 20 minutes are not convincing.
Interpretation of Results
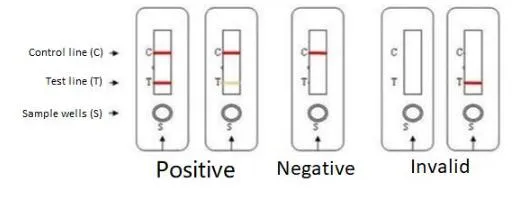
Negative
Only the C line is present. HIV-1/2 antibodies not detected.
Positive
Both C line and T line are present. Indicates presence of HIV-1/2 antibodies.
Invalid
Control (C) line does not appear. Result is invalid regardless of T line.
Performance Characteristics
- 🛡️ Minimum Detection: 0.2NCU/ mL
- 🛡️ Sensitivity: 100% positive detection rate for HIV-1 and HIV-2 reference samples.
- 🛡️ Specificity: 96.2% negative coincidence rate.
- 🛡️ Precision: 100% coincidence rate in repeated tests over 20 days.
- 🛡️ Subtypes Detected: HIV-1 (Type A, B, C, E, O) and HIV-2.
Limitations
1. Clinical auxiliary use only; results should be used with other clinical examinations.
2. Negative results do not rule out infection during the window phase.
3. Antiviral treatment may cause false negatives.
4. Only validated for urine samples; other body fluids may be inaccurate.


Frequently Asked Questions (FAQ)
What is the intended use of this kit?
It is designed for the qualitative determination of human immunodeficiency virus type I (HIV-1) and type II (HIV-2) antibodies specifically in human urine samples.
How long can I store the test kit?
The kit is valid for 24 months when stored at 4~30ºC in a dry environment and away from light.
How much urine is required for the test?
You need to add approximately 3 drops (about 100ul) of the urine sample to the sample well of the cassette.
When should I read the test results?
Results should be read exactly at 15 minutes. Any results read after 20 minutes are considered non-convincing.
Does a negative result completely rule out HIV infection?
No, a negative result does not rule out infection, particularly if the individual is in the "window phase" or undergoing antiviral treatment.
Can this test detect different HIV subtypes?
Yes, it can detect HIV-1 types A, B, C, E, and O, as well as HIV-2.