1 / 5










| Item | Details |
|---|---|
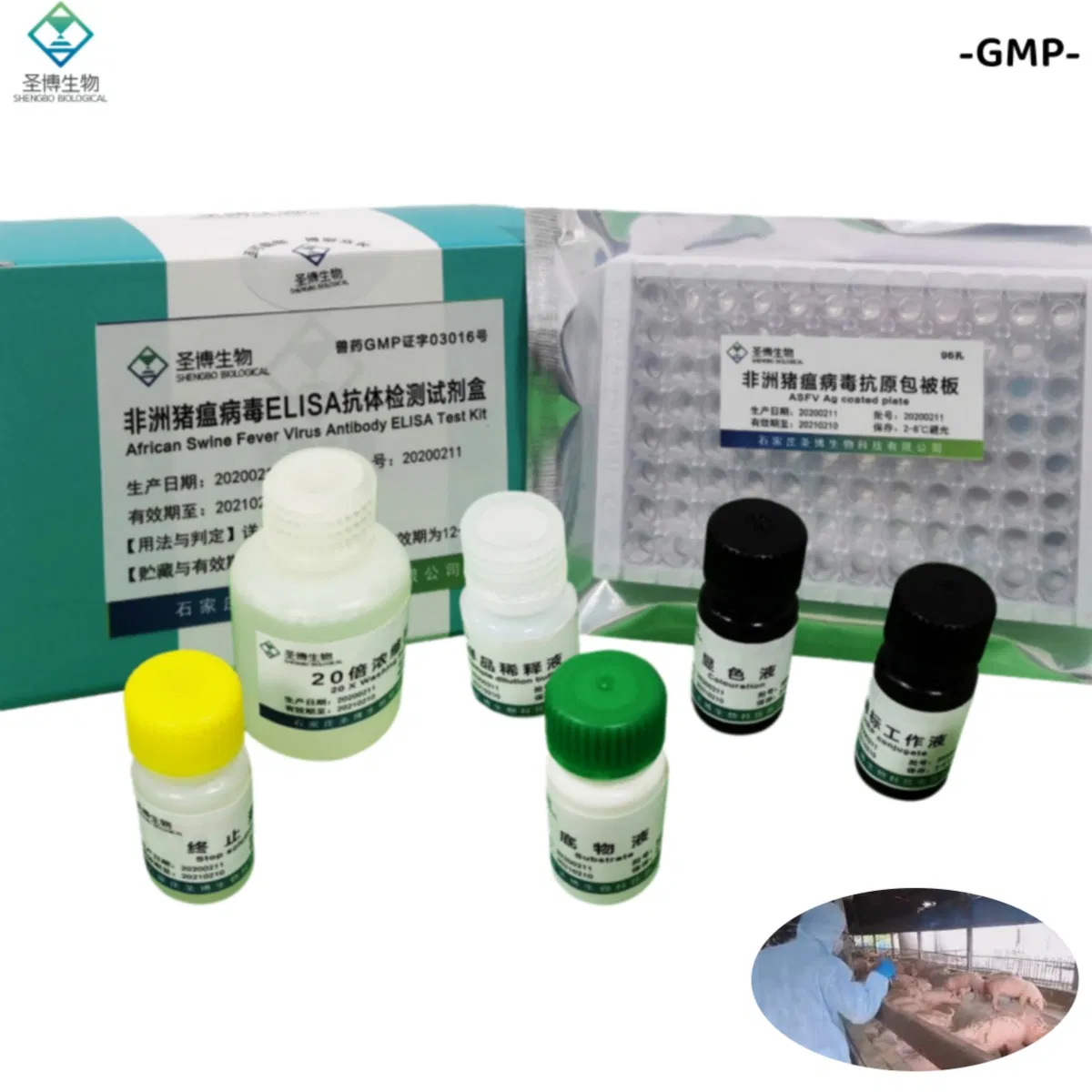
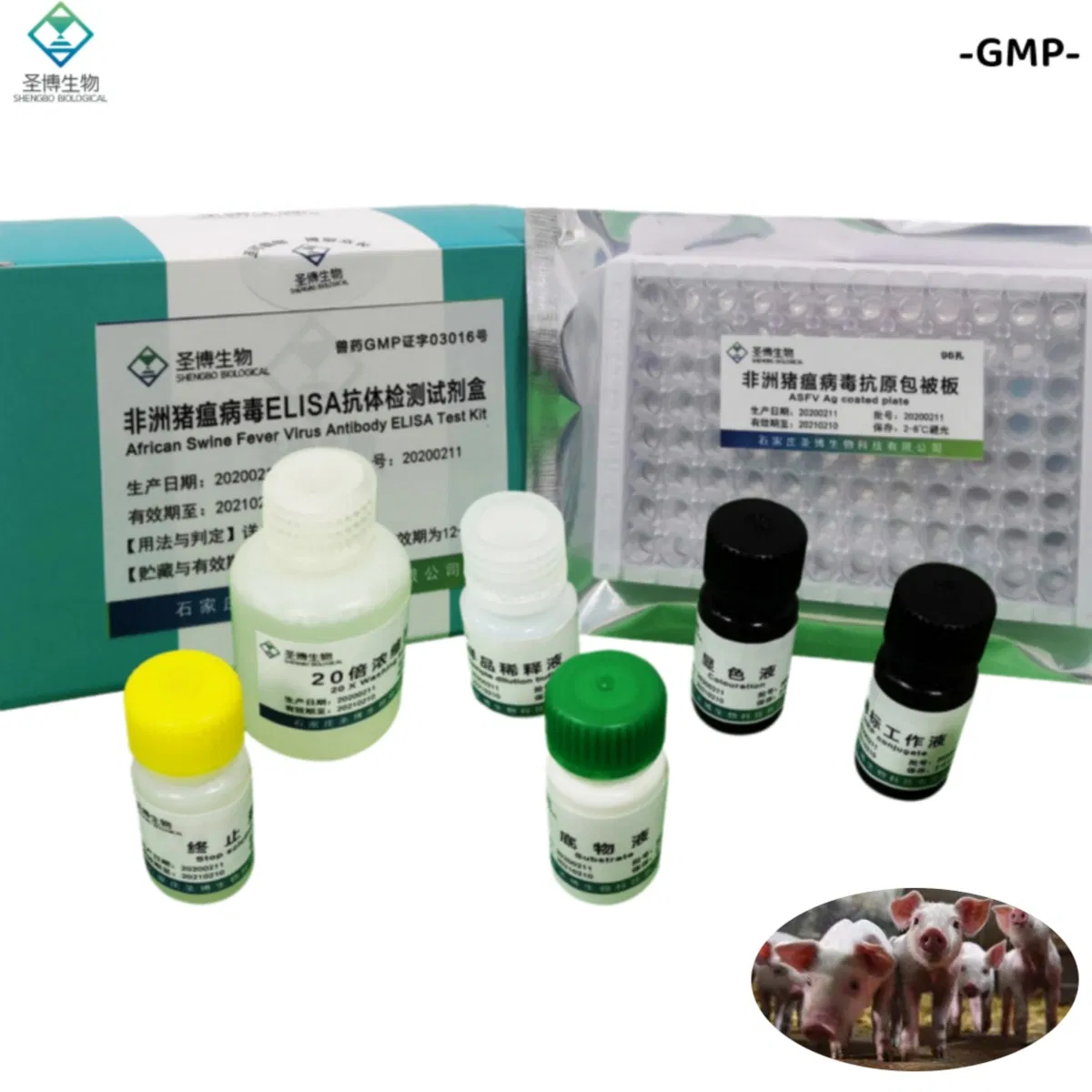
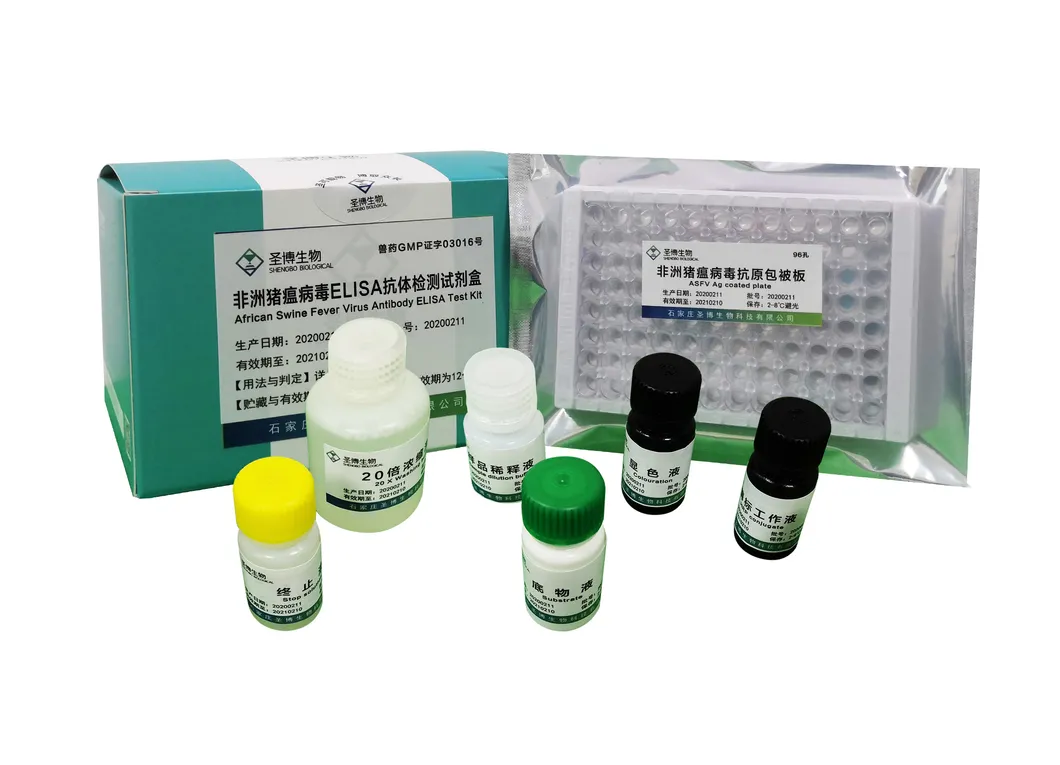
| Name | African Swine Fever Virus Antibody ELISA Test Kit |
| Package | 96 T/Box |
| Storage | 2~8ºC |
| No. | Name | 96T×1 | 96T×2 |
|---|---|---|---|
| 1 | Enzyme working reference solution | 11mL | 22mL |
| 2 | Sample diluted solution | 11mL | 22mL |
| 3 | Positive reference serum | 1mL | 2mL |
| 4 | Negative reference serum | 1mL | 2mL |
| 5 | Substrate solution | 6mL | 12mL |
| 6 | Color solution | 6mL | 12mL |
| 7 | Stop solution | 6mL | 12mL |
| 8 | 20× concentrated lotion | 50mL | 100mL |
| 9 | Instruction | 1 piece | 1 piece |
| 10 | Sealing film | 3 pieces | 6 pieces |
Established in 1998, our facility covers over 26,000 square meters. In 2015, eleven workshops and fourteen production lines received the GMP Certificate. We are currently one of the most comprehensive manufacturers of prepared drugs in China, with products exported to America, Africa, Europe, the Middle East, and South Asia.



We have invested over 1 million yuan in quality management hardware, including high-performance liquid chromatographs, UV-visible spectrophotometers, and bacterial endotoxin analyzers to ensure international quality standards.





