













(Fluorescence RT-PCR Method)
This kit is used for the detection of Rotavirus (Group A, B, C) Nucleic Acid and assisted diagnosis and epidemiological surveillance of Rotavirus (Group A, B, C).
| Number | Composition | 50 tests / kit | 200 tests / box |
|---|---|---|---|
| 1 | PCR Reaction Solution | 750μL × 1 tube | 750μL × 4 tube |
| 2 | Enzyme Mixture | 250μL × 1 tube | 250μL × 4 tube |
| 3 | Negative Control | 100μL × 1 tube | 100μL × 4 tube |
| 4 | Positive Control | 100μL × 1 tube | 100μL × 4 tube |
| 5 | Instructions | 1 serving | 1 serving |
Rotavirus is one of the main pathogens that cause diarrhea in infants and young children. It mainly infects small intestinal epithelial cells, causing cell damage and diarrhea. Rotavirus is prevalent in summer, autumn and winter every year. The route of infection is fecal-oral route.
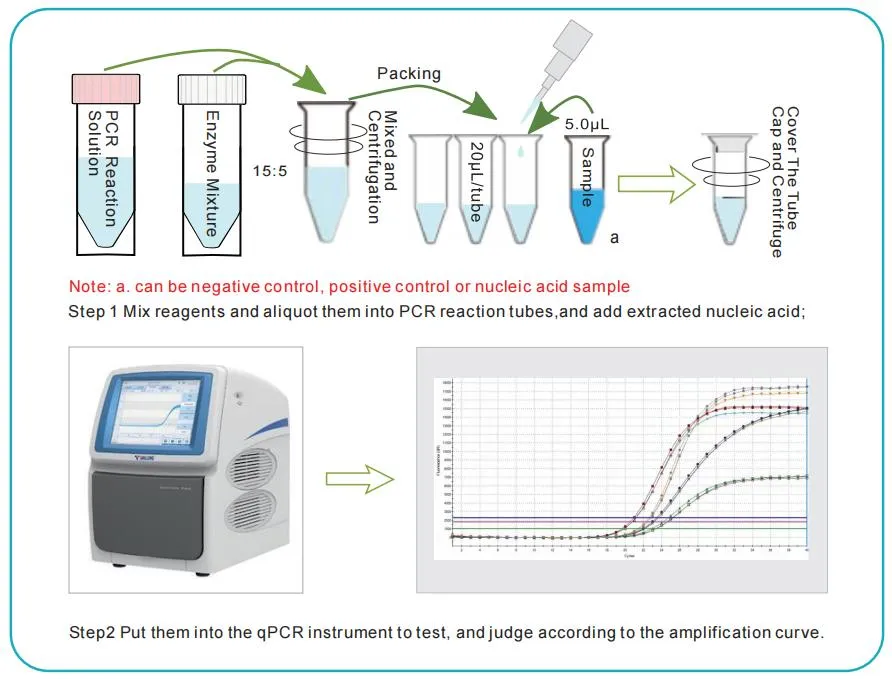
This kit is based on the principle of fluorescent PCR technology, using specific primers and Taqman probes for Rotavirus (Group A, B, C). The detection system includes internal control primers and probes to monitor sample collection and extraction, avoiding false negative results.
Storage Conditions: The kit should be stored at -20 ± 5°C. The expiry date is set at 12 months. Please use the reagent within 1 month after opening. Avoid freezing and thawing more than 5 times.
Applicable Instruments: Suitable for ABI series, Bio-Rad series, Agilent Stratagene MX series, Roche LightCycler R480, Cepheid SmartCycler, Rotor-Gene series, and other multi-channel real-time quantitative PCR instruments.

Founded in 2018, the manufacturer is based in the fields of molecular biology and diagnostics, utilizing a unique "product + service" model. The R&D team consists of worldwide experts, operating within high-standard 100,000-grade GMP production workshops and holding ISO9001 quality management system certification.



