










This product is used for the qualitative determination of human immunodeficiency virus type I (HIV-1) antibodies and type II (HIV-2) antibodies in human urine samples. It is suitable for the auxiliary diagnosis of HIV infection. The test results are only for clinical reference and cannot be used alone as the basis for confirming or excluding cases. In order to achieve the purpose of diagnosis, the test results should be used in conjunction with clinical examination, medical history and other examinations. This product can be used for consumer self-test.
| Specifications | Details |
|---|---|
| Product Name | HIV Antibody Urine Test kit (Colloidal Gold Method) |
| Method of Detection | Colloidal Gold Method |
| Qualitative/Quantitative | Qualitative |
| Specimen | Urine |
| Required Volume | 3 drops (approximately 100ul) |
| Packaging | Cassette: 1 test/box, 10 tests/box, 20 tests/box, 50 tests/box |
Allow the test box to equilibrate to room temperature prior to testing.
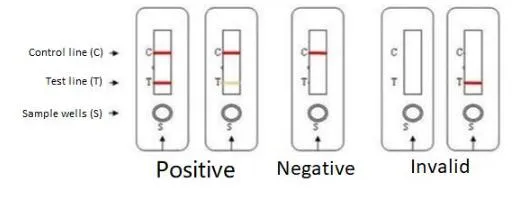
Negative: Only the C line is present. HIV-1/2 antibodies not detected.
Positive: Both C-line and T-line are present. Indicates presence of HIV-1/2 antibodies.
Invalid: Control line (C) does not appear. The test must be repeated.
1. Minimum detection amount: 0.2NCU/ mL.
2. Sensitivity: 100% positive detection rate for HIV-1 and HIV-2 reference samples.
3. Specificity: 96.2% negative coincidence rate in clinical urine samples.
4. Precision: 100% coincidence rate in continuous testing over 20 days.
5. Cross reaction: No cross reaction with Syphilis, Hepatitis B/C, Flu A/B, COVID-19, etc.
6. HIV subtypes detected: Detects HIV-1 (type A, B, C, E, O) and HIV-2.



