Company Overview
An international high-tech enterprise driven by innovation in the area of medical devices R&D, production and sales. The focus is on diagnosis of microorganisms, tumors and some rare diseases, mainly engaged in the research and development, production and sales of in vitro diagnostic reagents and automatic instruments.
With R&D centers in China and the US, the enterprise holds independent intellectual property rights for fluorescent staining, protein labeling, and time-resolved immunofluorescence detection technologies. The facilities include over 10,000 square meters of GMP purification production workshops meeting FDA, CE, and CFDA standards.
Workshop & Exhibition
Frequently Asked Questions
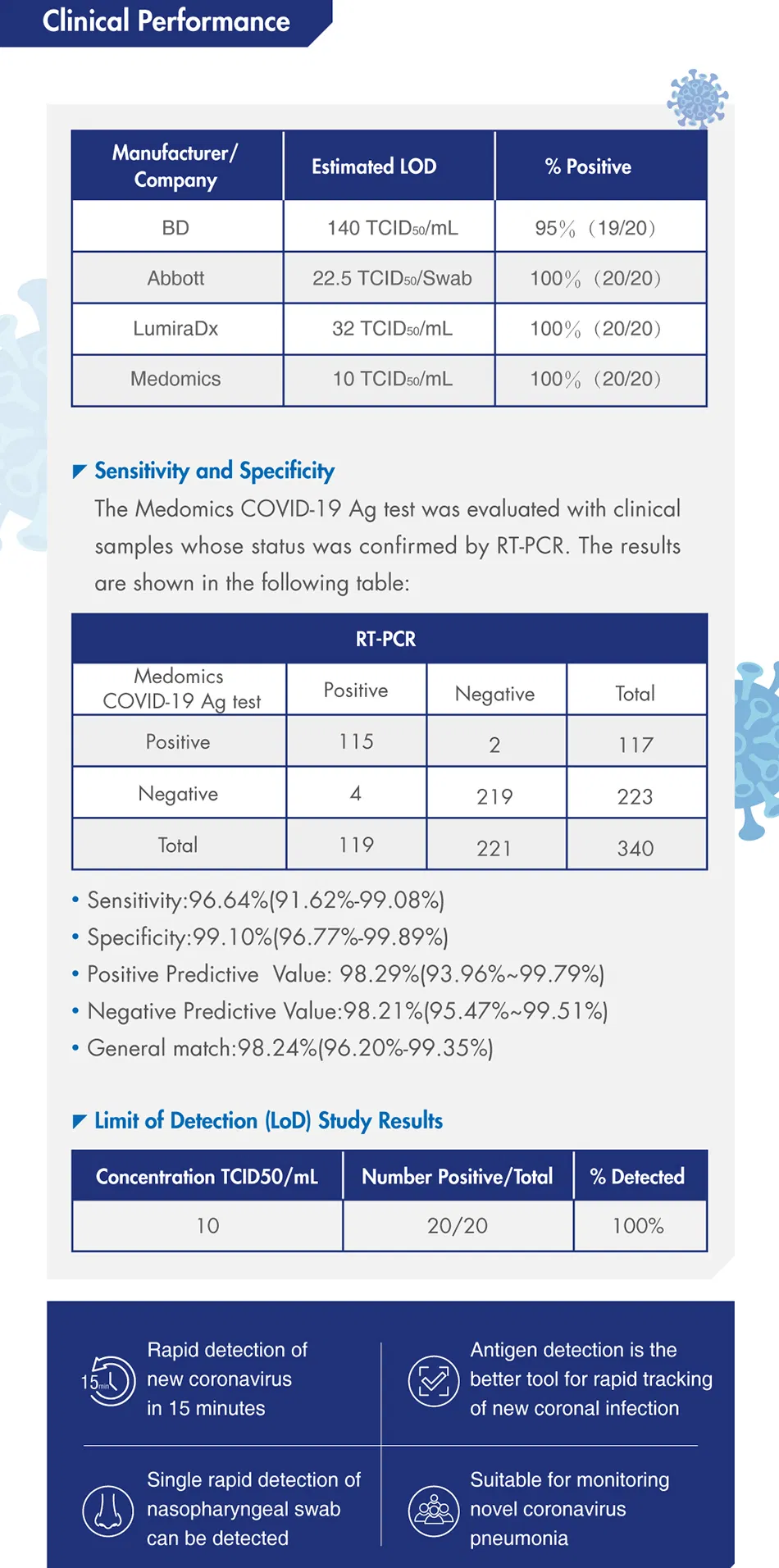
What is the accuracy of this Rapid Antigen Test?
The test demonstrates high clinical performance with a sensitivity of 96.64% and a specificity of 99.10%.
How long does it take to get the results?
The reaction time is very efficient, providing clear results within 15 to 20 minutes.
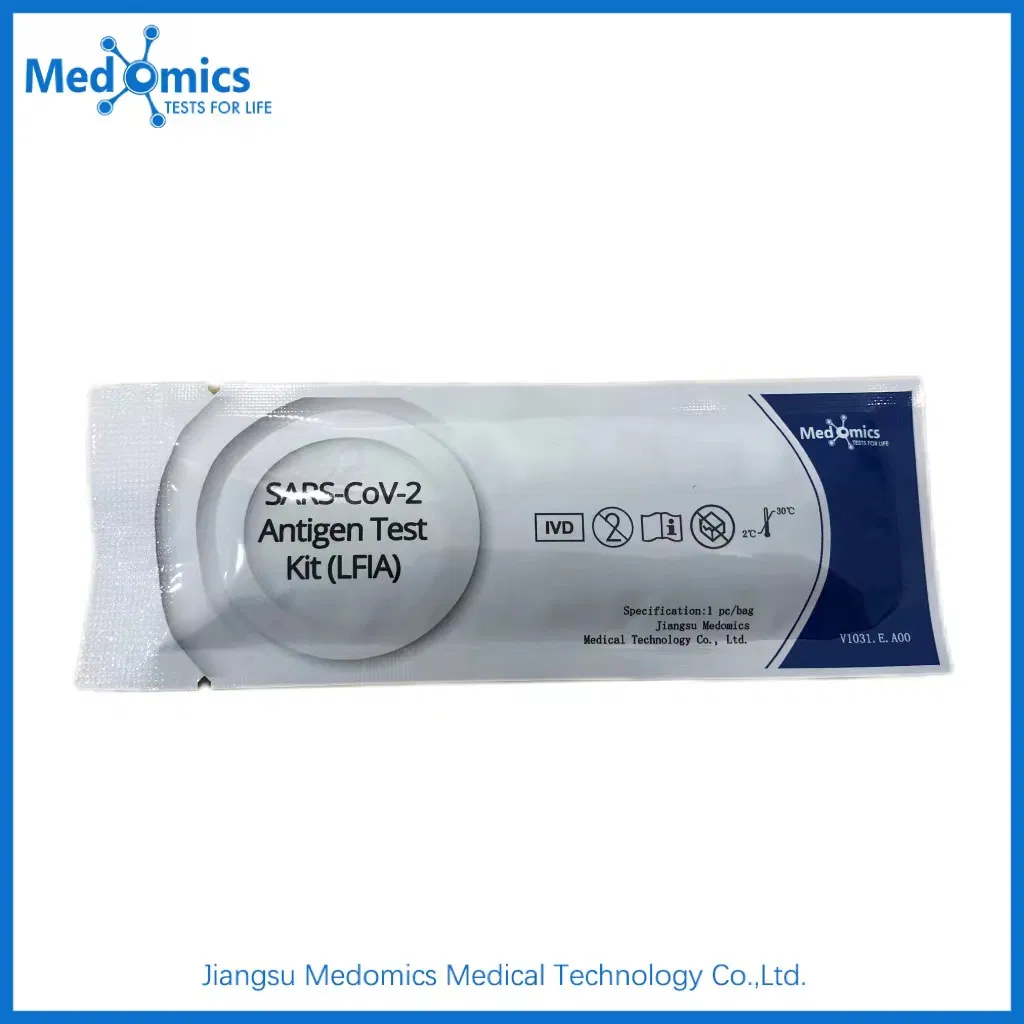
What are the storage requirements for the kits?
The kits should be stored in a cool, dry place at a temperature range between 2°C and 30°C.
Does the product have international quality certifications?
Yes, the product is fully certified with CE and ISO13485, ensuring it meets international medical device standards.
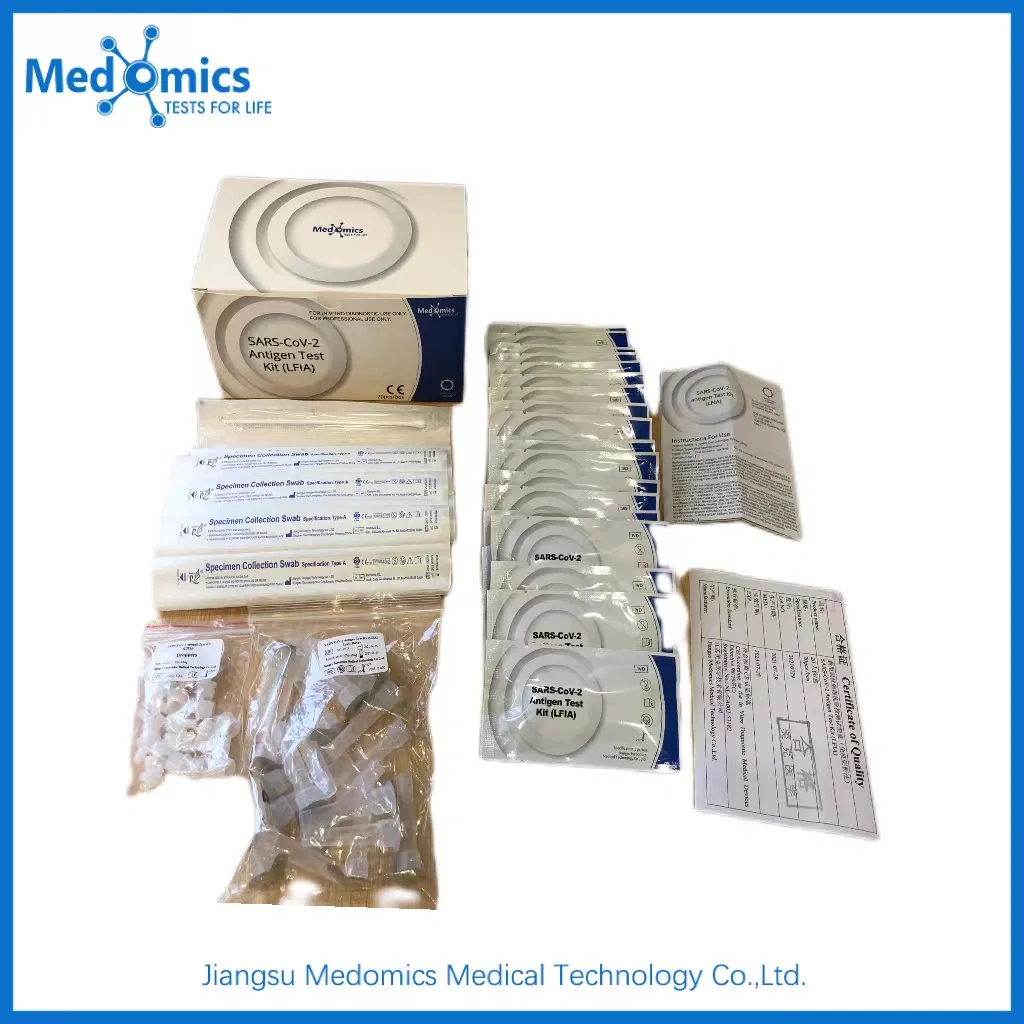
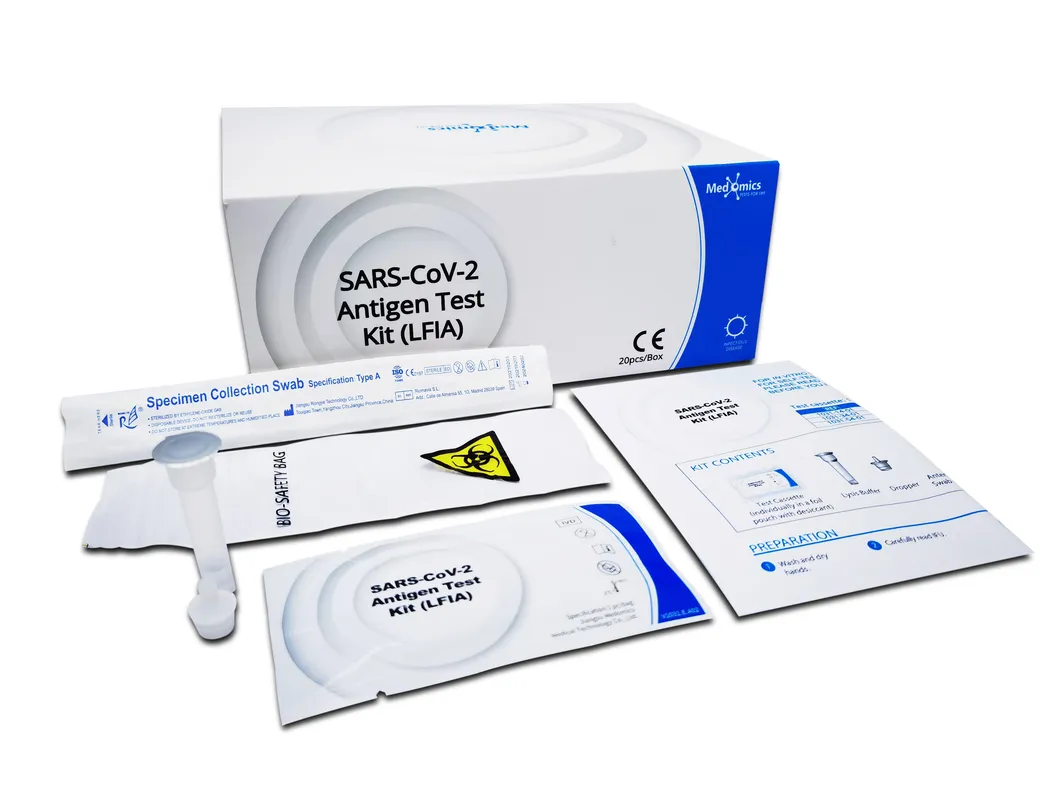
What is the standard packaging for bulk orders?
The standard kit contains 20 tests. For shipping, these are packed in boxes and then into sturdy cartons to ensure safety during transport.
Can you handle large volume monthly orders?
Yes, our production facility has a capacity of approximately 5,000,000 pieces per month to support global demand.