



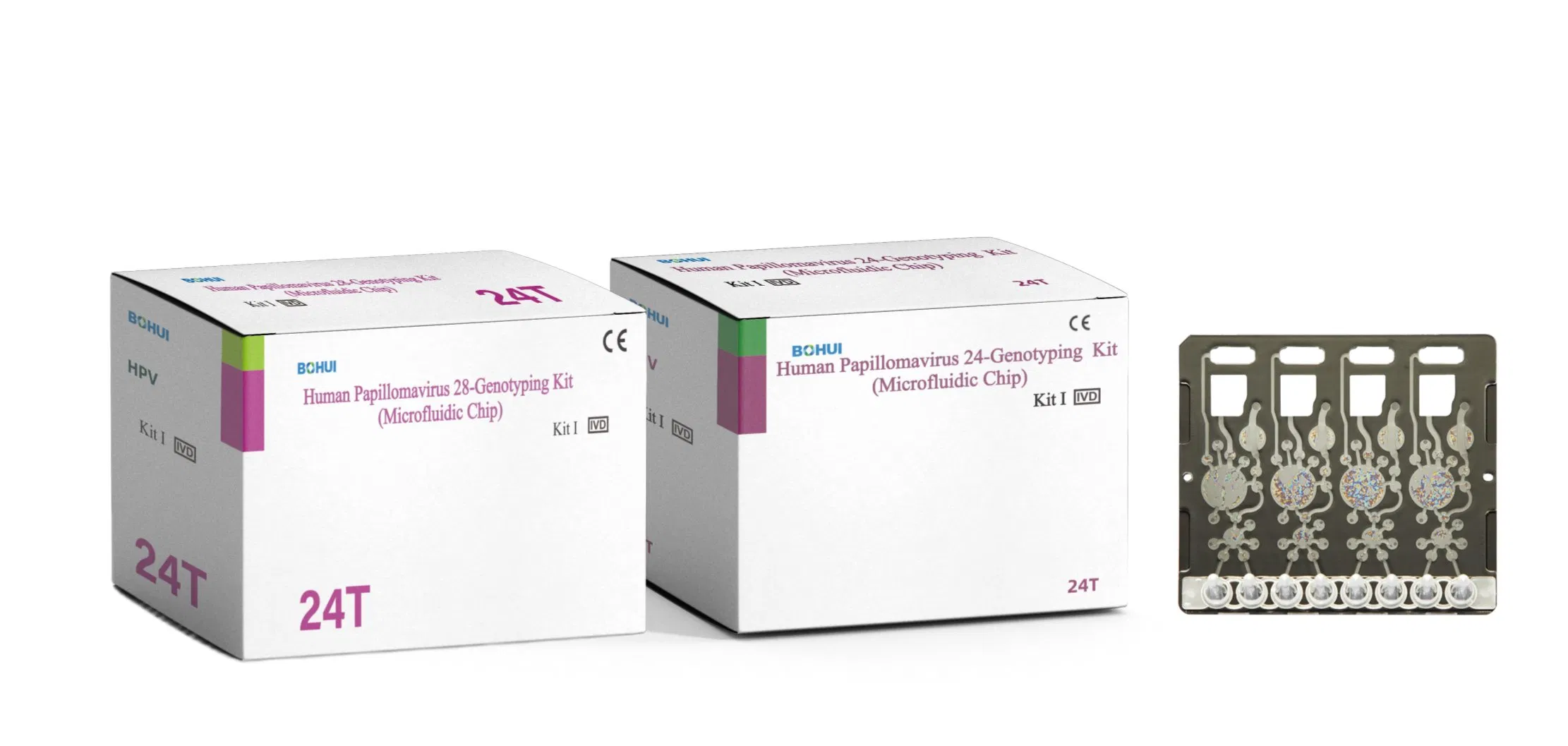










The instrument uses reverse hybridization assay for viral sub-type analysis. When specimens are placed in the slot, the instrument automatically adds necessary reagents, extracts DNA, and amplifies PCR via micro-pumps on the Chip. Amplified products are transferred to the test area for reverse hybridization with probes. If target gene is present, it is captured by the probe, leading to coloration and identification of the virus sub-type. Negative specimens show no spots on probe positions.
Nucleic Acid Chip Analyzer comprises a host unit and optional printer.
Host Unit Integrates:
Sample Type: Exfoliated cervical epithelial cells
Application: Ideal for nucleic acid detection and clinical screening like HPV testing.



| Parameter | Specification |
|---|---|
| Model | BHF-VI |
| Detection Platform | PCR & Reverse dot blot hybridization |
| Detection Modules | 6 Modules |
| Sample Capacity | 24 |
| Heating rate | ≥1.5 ℃/s (from 50℃ to 90℃) |
| Cooling rate | ≥1.5 ℃/s (from 90℃ to 50℃) |
| Electrical specification | 230V, 50Hz, 600VA |
| Dimension | 1000 x 610 x 690±10 (mm) |
| Altitude | ≤2300m |
| Weight | ≤80kg |
5 minutes of hands-on time
No precise measuring or pipetting needed
Turnaround time of around 3.5 hours
24/28 target genotypes HPV panel



Founded in 2001, the group is a high-tech biomedical enterprise integrating R&D, production, sales, and service. It boasts a 40,000-square-meter R&D center and is a publicly listed entity known for its innovation in the field of in-vitro diagnosis.
The enterprise showcases robust independent R&D capabilities with a dedicated team. Through continuous innovation, it has developed platforms like molecular diagnosis, immunoassay, atomic absorption, and mass spectrometry. The fully automated nucleic acid testing products, based on microfluidic technology, are lauded as a "laboratory on a chip" and serve numerous medical institutions globally.

