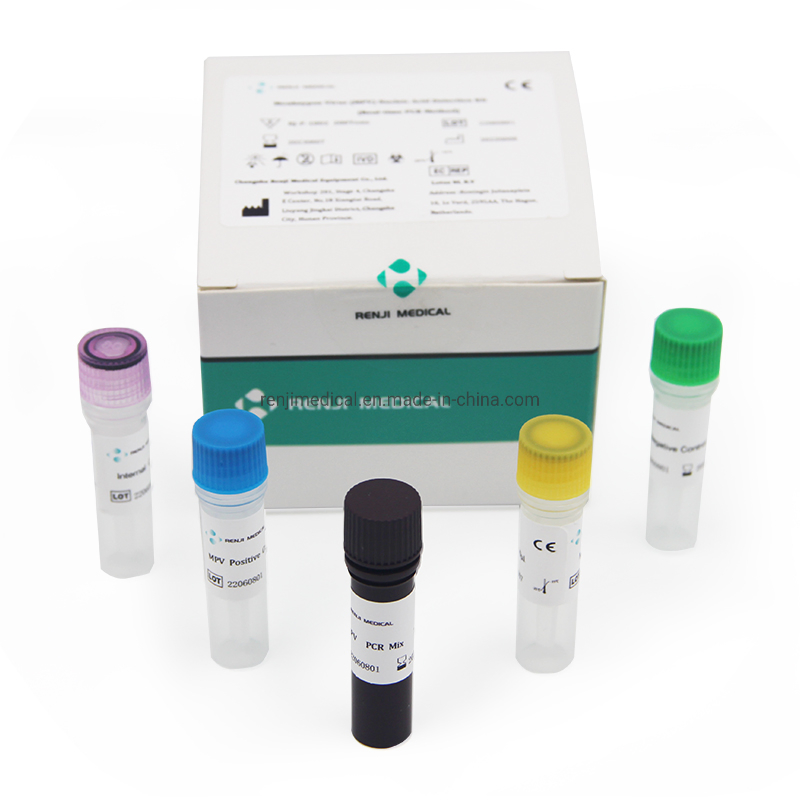
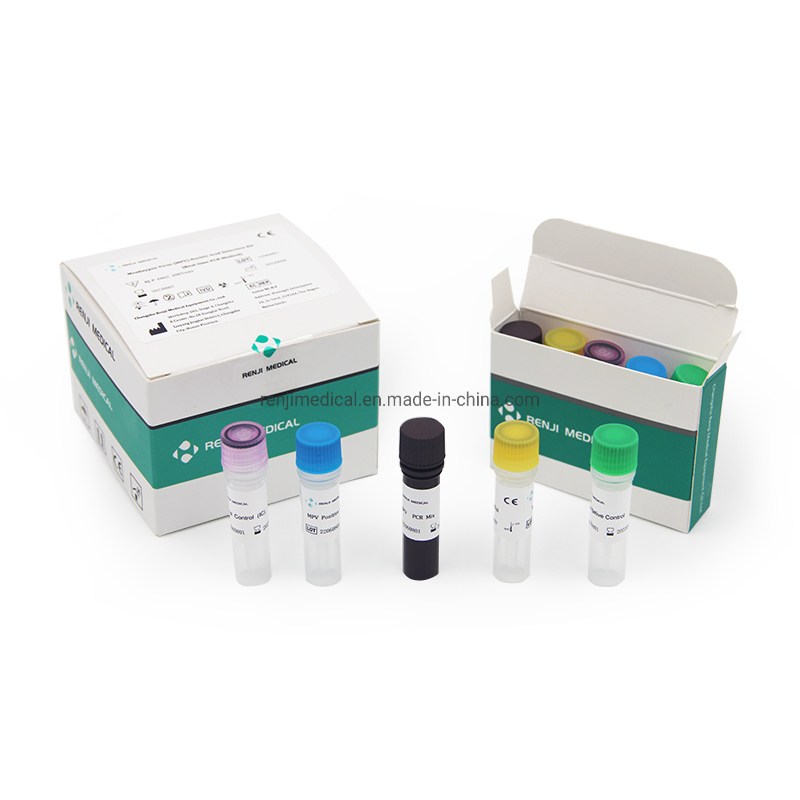

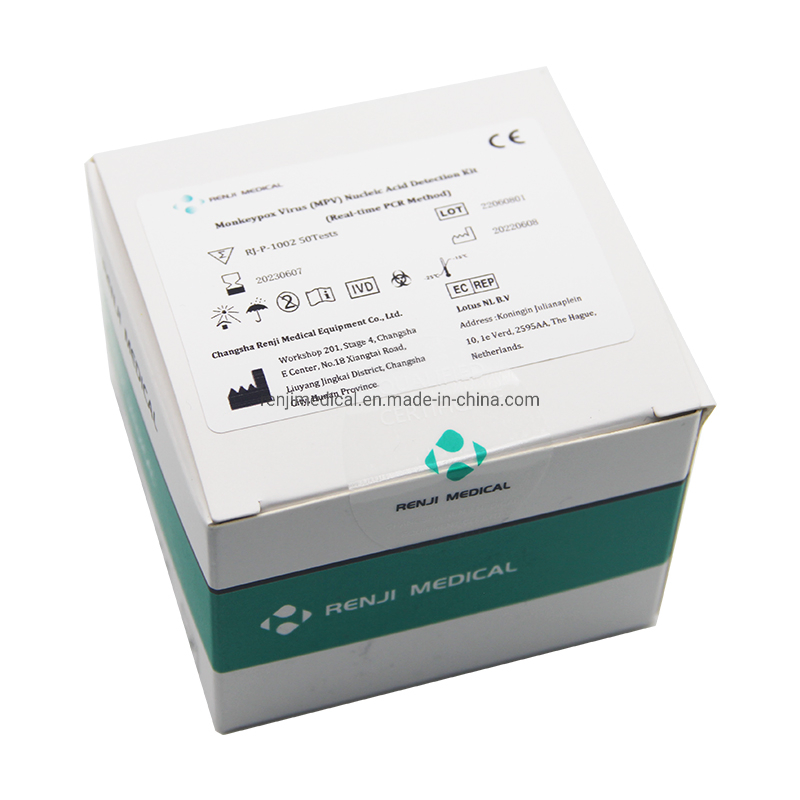











Monkey pox is a zoonotic virus, causing symptoms similar to small pox, though less severe clinically. It belongs to orthopoxvirus genus of Poxviridae family. Two clades of Monkeypox are known: the West African and the Congo Basin (Central African). It can be transmitted from one person to other through close contact with lesions, body fluids, droplets and contaminated material such as linen. The incubation period for infection is usually 6-13 days, however it can vary from 5 to 21 days.
As per WHO, possible symptoms may include headache, fever, swollen lymph nodes, weakness, muscle and body ache. The Monkeypox virus can be detected by real time polymerase chain reaction, transmission electron microscopy and immunohistochemistry.
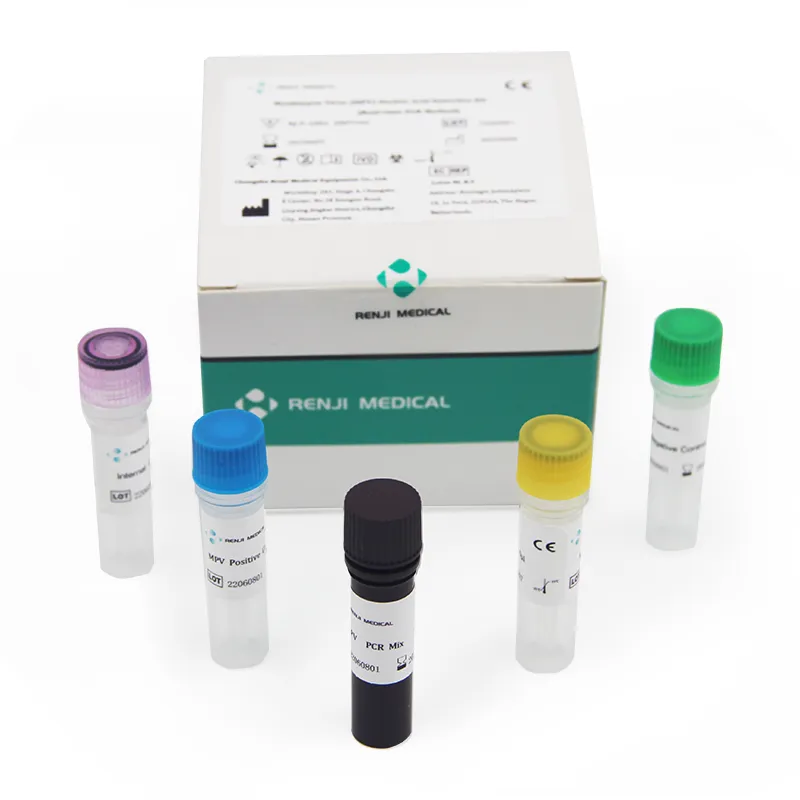
The Monkeypox Virus Real Time PCR kit is used for the detection of Monkeypox virus in serum or lesion exudate samples by using real time PCR systems. We focus on providing high-quality medical diagnostic solutions for global health needs.
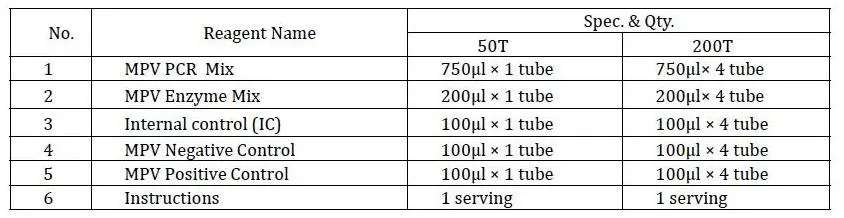
Equilibrate reagents at room temperature, vortex each component. Prepare PCR-Mastermix (PCR Mix 15 μl + Enzyme Mix 4 μl + IC 1 μl per test) and centrifuge.
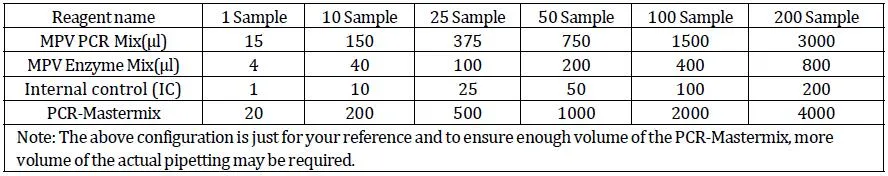
Extract viral DNA using recommended extraction kits. Add 20μl PCR-Mastermix into PCR reaction tube with 5μl processed sample, Positive Control, and Negative Control.
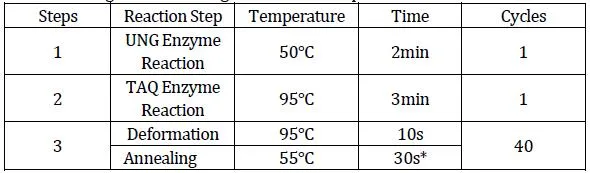
Set cycle parameters according to the standardized amplification protocols for fluorescence quantitative PCR instruments.

Our production facilities maintain strict quality control standards. As a direct manufacturer, we ensure all medical consumables meet international requirements.





