




In the modern industrial era, the role of a Laboratory Management Software Manufacturer has shifted from providing simple database tools to delivering comprehensive, AI-integrated digital ecosystems. Globally, laboratories in the pharmaceutical, clinical, and environmental sectors are undergoing a massive "Digital Lab" transformation. The shift from manual record-keeping to Laboratory Information Management Systems (LIMS) is no longer a luxury but a fundamental necessity for data integrity and operational efficiency.
The global LIMS market is projected to reach billions in value by 2030, driven by the increasing need for regulatory compliance (such as FDA 21 CFR Part 11 and ISO 17025) and the demand for high-throughput testing. As a leading manufacturer based in China, we recognize that the synergy between software logic and hardware precision—like our high-resolution imaging and automated analysis systems—is the key to unlocking true laboratory potential.
The future of laboratory management is defined by "Hyper-Automation." We are at the forefront of this evolution, integrating several key trends into our software and hardware solutions:
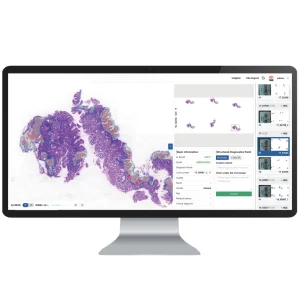
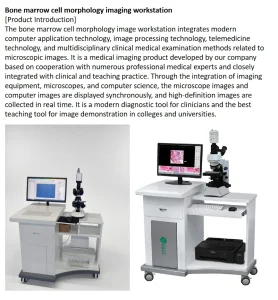
Utilizing machine learning algorithms to assist in pathology diagnosis and cell morphology analysis, reducing human error and accelerating turnaround times.
Modern labs require real-time data access. Our systems offer secure cloud storage, allowing multi-location labs to synchronize data instantly across the globe.
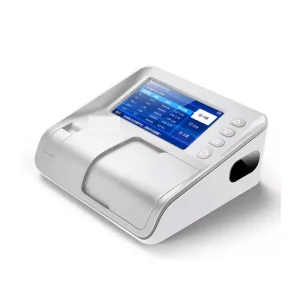
Every piece of hardware, from centrifuges to biochemical analyzers, is designed to connect seamlessly with the management software for automated data logging.
Global enterprises today look for more than just a software vendor; they seek a strategic partner. When procuring laboratory management systems, organizations prioritize Interoperability, Security, and Localization.
Our solutions are designed with "Global Logic, Local Application" in mind. Whether it's a diagnostic center in Europe requiring strict GDPR compliance or a clinical lab in Southeast Asia needing multi-language support and affordable consumables, our platforms are customizable. We understand that a hospital in Brazil has different localized workflow requirements than a research institute in Germany. Our engineering team provides modular software updates that adapt to these specific regional medical regulations and cross-cultural communication needs.
| Procurement Factor | Global Standard Requirement | Our Factory Solution |
|---|---|---|
| Data Integrity | FDA 21 CFR Part 11 / ALCOA+ | Full Audit Trails & Encrypted Logs |
| Integration | HL7 / LIS / ERP Compatibility | Open API & Custom Middleware |
| Cost Control | Scalable Licensing & Maintenance | Tiered Pricing & Low-cost Consumables |
China has become the global hub for medical technology innovation. As a premier manufacturer, we offer unique advantages that Western counterparts often cannot match:
We manufacture both the hardware (Centrifuges, Analyzers, Scanners) and the software. This ensures 100% compatibility and eliminates the "software-hardware gap" that causes system crashes.
Located in the heart of the global electronics supply chain, we can prototype and iterate software features or hardware components in weeks, not months.
Leveraging lean manufacturing and large-scale production allows us to provide high-end diagnostic technology at a fraction of the traditional market price.
Professional Insight: "As a professional medical device supplier based in China, we adhere to the philosophy of 'Quality First, Customer Supreme.' Backed by lean manufacturing capabilities and strict quality control systems, we are dedicated to building long-term partnerships and delivering dependable healthcare solutions to customers worldwide."
We specialize in the manufacturing and global distribution of a wide range of medical products, including hospital equipment, medical disposables, surgical dressings, laboratory products, and home healthcare solutions. With a strong commitment to the principle of “quality first, customer foremost,” we have earned the trust and recognition of clients across international medical markets.

Our product portfolio is designed to meet the diverse needs of hospitals, clinics, laboratories, and distributors, ensuring reliable performance, safety, and compliance with international standards. From essential medical consumables to advanced diagnostic and laboratory support products, we strive to deliver consistent quality and value.

Our experienced foreign trade team possesses in-depth knowledge of international trade procedures, regional medical regulations, and cross-cultural communication. This enables us to provide efficient and professional support throughout every stage of cooperation. We aim to be a true one-stop supplier, offering not only a comprehensive range of medical products but also integrated services including consultation, sourcing, order tracking, and after-sales support.