














| Basic Information | |
|---|---|
| Certification | CE, FDA, GMP, ISO |
| Control System | Fully Automatic |
| Treatment Process | Filtration, Ultrafiltration |
| Warranty of core components | 1 Year |
| Power Supply | 380V 50Hz |
| Tank Material | Stainless Steel 316L |
| Weighing Control Accuracy | 3% |
| Sealing and Protection Levels | IP65 |
| Temperature Control Accuracy | -10~150ºC ±0.2ºC |
| Stirring Speed Control Accuracy | 0~450rpm ±3.0% |
| Transport Package | Wooden Box |
| Origin | China |
The Pharmaceutical Formulation System is a specialized apparatus designed for liquid mixing and dispensing operations. This equipment enables the preparation of pharmaceutical solutions by combining active pharmaceutical ingredients (APIs) with solvents such as water for injection. Through integrated processes including controlled stirring, heating, sterilization, and filtration, the system produces medicinal solutions at precise target concentrations. These systems find application across both sterile and non-sterile pharmaceutical manufacturing environments.


Pre-tested modular equipment reduces installation costs, accelerates delivery, and ensures flexibility.

1-click CIP/SIP with auto-detection. Features include recipe management, e-signature, e-records, and audit trails.
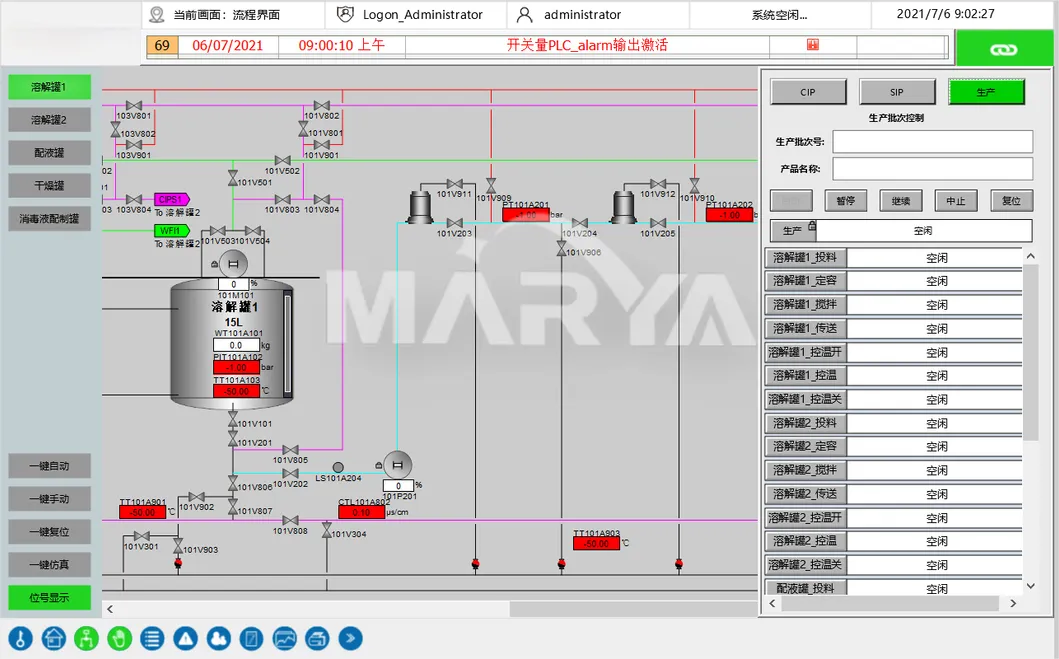
Critical parameter control (temp, DO, pH) with auto-alerts for deviations to ensure drug quality.

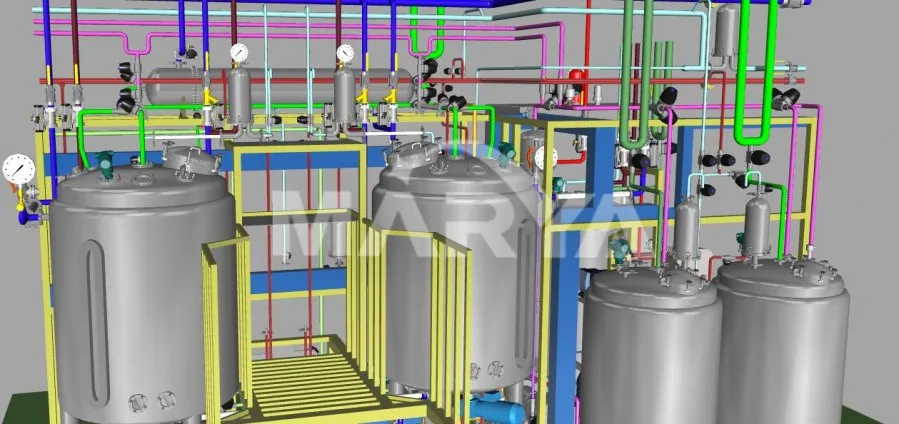
Pre-production 3D modeling optimizes piping, valve, and instrument placement for user-friendly operation.
| Name | Parameters |
|---|---|
| Working volume | 2~15000L |
| Material | 304, 316L, 904L, TA2 |
| Stirring type | Upper mechanical, lower magnetic stirring |
| Stirring speed accuracy | Speed range 0~450rpm ±3.0% |
| Temperature accuracy | Range -10~150ºC ±0.2ºC |
| Pressure accuracy | Range -0.01Mpa~0.06Mpa ±0.01MPa |
| Weighing accuracy | 3‰ |
| Protection levels | IP65 |










A: Our systems are highly scalable, with working volumes ranging from small-scale 2L units up to large industrial 15,000L tanks.
A: Yes, the system is designed to meet CE, FDA, GMP, and ISO standards, ensuring full compliance for sterile and non-sterile drug manufacturing.
A: The system features a 1-click CIP (Clean-in-Place) and SIP (Sterilize-in-Place) operation with automated detection to ensure thorough cleaning and sterilization completion.
A: All liquid contact parts are made from high-grade Stainless Steel 316L with electrolytic polishing (Ra < 0.4µm) to prevent contamination.
A: Yes, our Complex Formulation System is specifically designed to handle advanced pharmaceutical processes including suspensions, emulsions, liposomes, and microspheres.
A: Absolutely. The system includes computerized features like audit trails, electronic signatures, and records to facilitate regulatory compliance and audits.